Application of Multiplex Fluorescent Immunohistochemistry (mIHC) in the Evaluation of Organ Transplant Rejection

1. Literature Information
This literature analysis focuses on a pivotal clinical study investigating transplant rejection mechanisms in renal allograft biopsies. The research employed multiplex fluorescent immunohistochemistry (mIHC) based on Tyramide Signal Amplification (TSA) technology to analyze 45 kidney transplant patient samples, providing critical insights into antibody-mediated rejection (ABMR) and T cell-mediated rejection (TCMR).
2. Research Background
Organ transplantation is a life-saving intervention for end-stage organ failure, but immune rejection remains a major challenge. Traditional diagnostic methods such as conventional IHC and RT-PCR have limitations in simultaneously visualizing multiple cell types and their spatial interactions in situ. Multiplex fluorescent immunohistochemistry (mIHC) enables up to six-target and seven-color staining on a single tissue section via cyclic labeling with TSA technology, offering unprecedented advantages for studying multicellular interactions and high-throughput biomarker analysis.
While mIHC is widely applied in tumor microenvironment and drug efficacy evaluation, its potential in transplant rejection research is significantly underutilized. This study addresses this gap by applying mIHC to precisely characterize immune cell infiltration and spatial distribution in kidney transplant rejection.
3. Research Design & Methodology
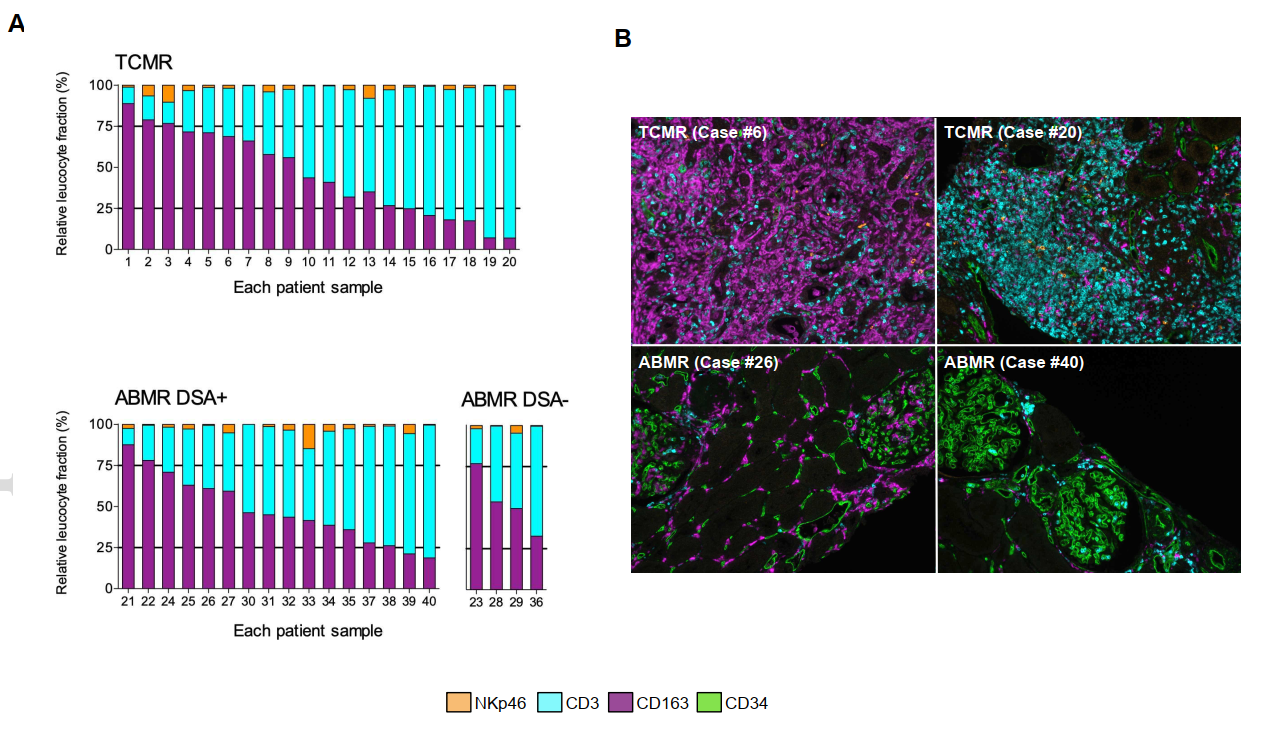
The study enrolled 45 kidney transplant recipients, stratified into three groups:
- 20 patients with antibody-mediated rejection (ABMR)
- 20 patients with T cell-mediated rejection (TCMR)
- 5 patients with no evidence of rejection (control group)
Using TSA-based mIHC, researchers performed four-marker, five-color staining on formalin-fixed paraffin-embedded (FFPE) kidney biopsy tissues, targeting NKp46, CD3, CD34, and CD163 to identify natural killer cells, T lymphocytes, vascular endothelial cells, and macrophages respectively. The research team validated mIHC results against conventional IHC (NKp46) and RT-PCR (CD3, CD163) to confirm methodological accuracy and reliability.
4. Key Research Findings
4.1 Methodological Validation
mIHC results demonstrated excellent consistency with conventional IHC and RT-PCR data, confirming the high reliability, specificity, and intuitive visualization advantages of the mIHC platform for transplant rejection analysis.
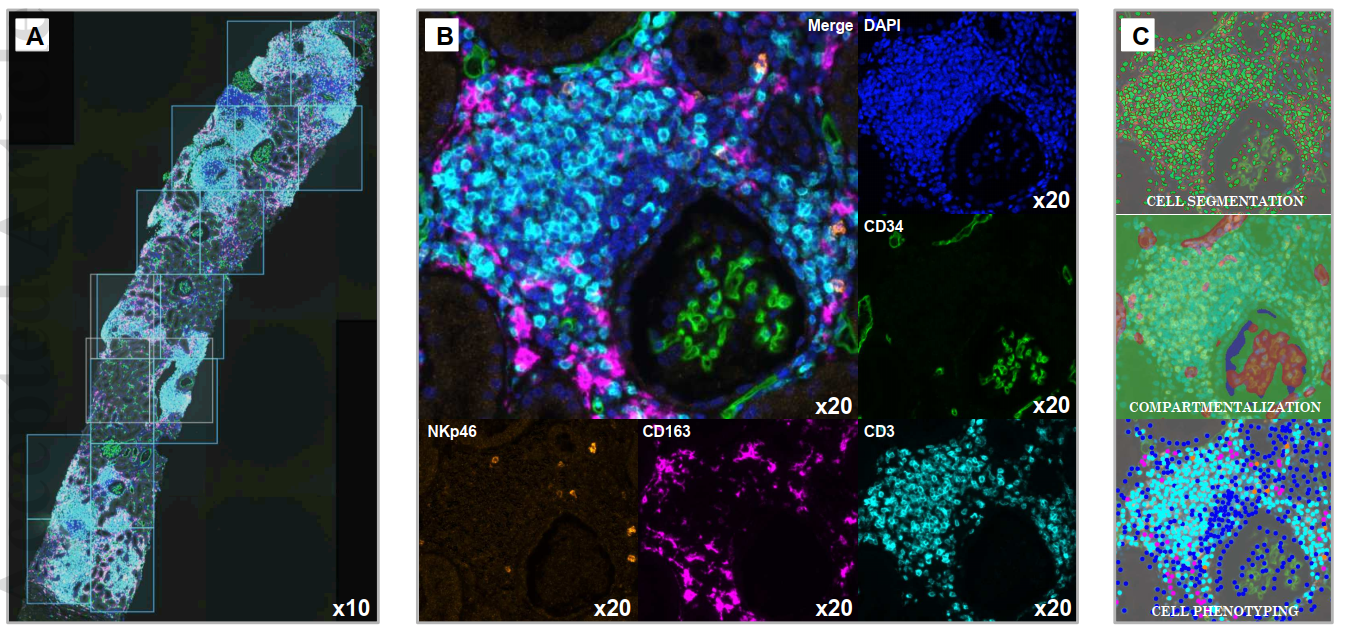
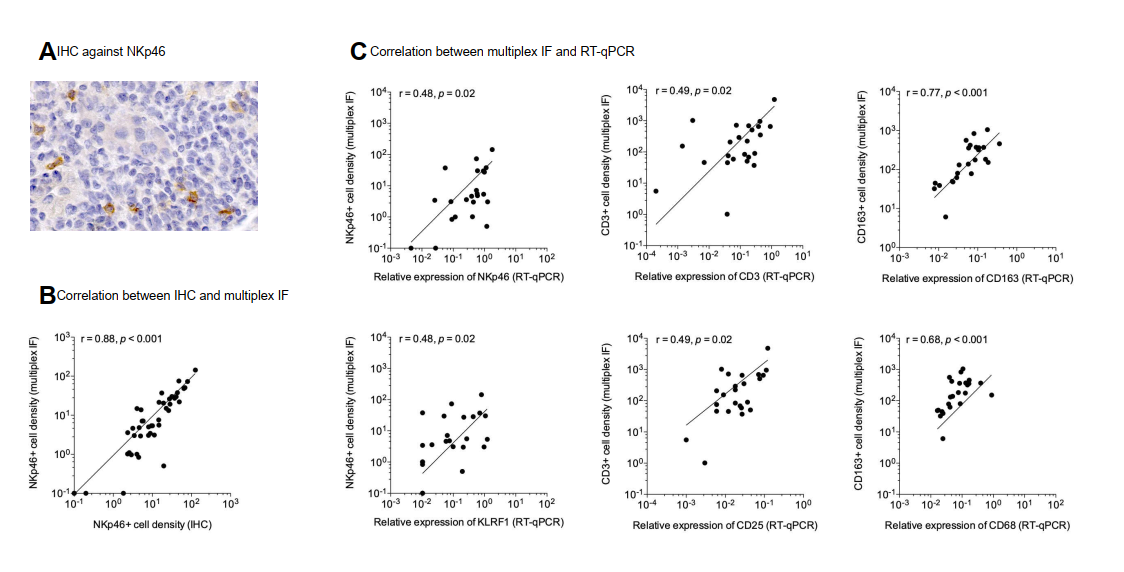
4.2 Immune Cell Infiltration Patterns
Significant elevation of CD34+ leukocytes, CD3+ T cells, CD163+ macrophages, and NKp46+ NK cells was observed in both ABMR and TCMR groups compared to the non-rejection control group. Notably, the TCMR group exhibited more robust immune cell recruitment.

4.3 Spatial Distribution Characteristics
mIHC enabled high-precision spatial analysis of infiltrating immune cells:
- In TCMR specimens, immune cells predominantly accumulated in the perivascular and interstitial compartments
- In ABMR specimens, immune cell infiltration was primarily localized within glomerular structures
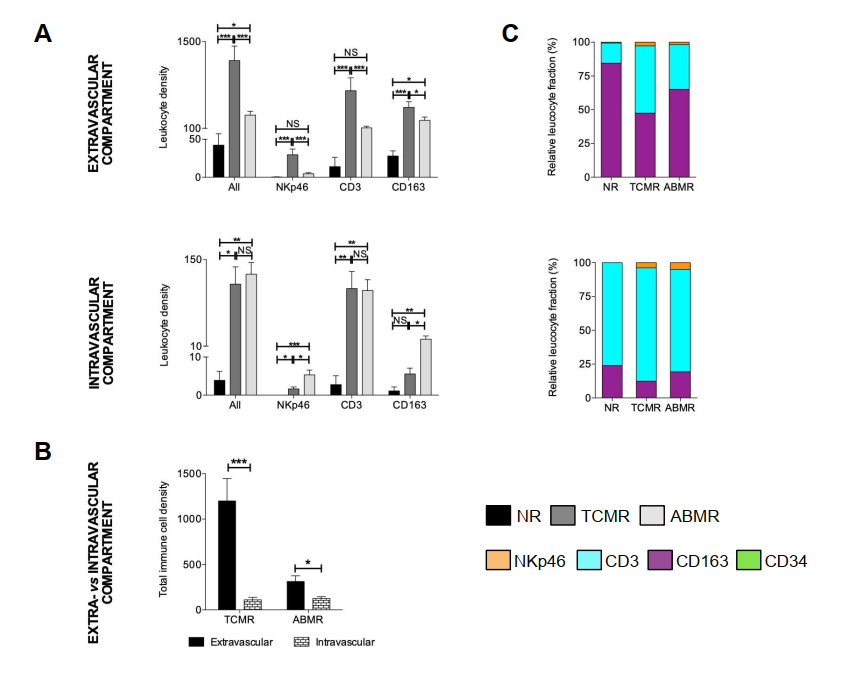
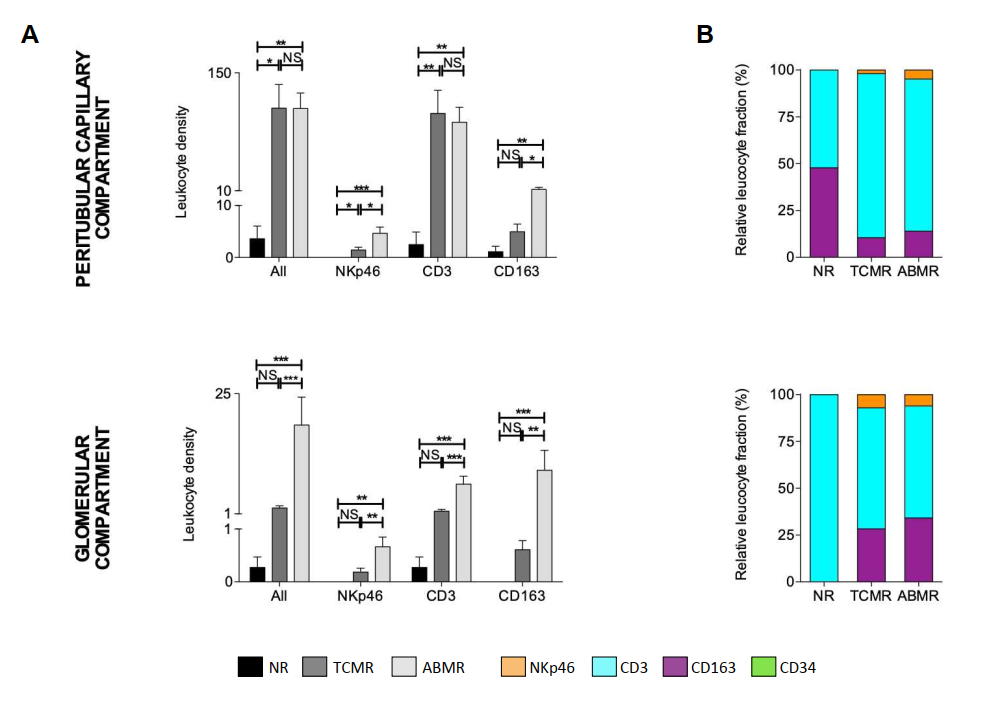
4.4 Individual Heterogeneity
The study further revealed substantial inter-individual variability in renal transplant rejection profiles, which can be effectively captured and quantified by mIHC technology.
5. Product Empowerment by ANT BIO PTE. LTD.
The high-quality multiplex fluorescent IHC reagents and kits developed and supplied by ANT BIO PTE. LTD. were instrumental in the success of this research. Our mIHC solutions leverage advanced TSA signal amplification technology to deliver ultra-sensitive, high-resolution multiplex staining, enabling simultaneous detection of multiple biomarkers on a single tissue section.
These products supported the researchers in achieving clear, specific, and reproducible staining results for NKp46, CD3, CD34, and CD163, facilitating accurate identification and spatial mapping of immune cell subsets. The robust performance of ANT BIO PTE. LTD. reagents ensured reliable data acquisition and comparative analysis across different patient groups, directly contributing to the discovery of distinct immune cell localization patterns in ABMR and TCMR.
6. Related High-Quality Reagents from ANT BIO PTE. LTD.
| Catalog No. | Product Name | Application | Sub-Brand |
|---|---|---|---|
| abs50012 | 4-Color Multiplex Fluorescent IHC Kit (Mouse-Rabbit Universal Secondary Antibody) | mIHC | Absin |
| abs50013 | 5-Color Multiplex Fluorescent IHC Kit (Mouse-Rabbit Universal Secondary Antibody) | mIHC | Absin |
| abs50014 | 6-Color Multiplex Fluorescent IHC Kit (Mouse-Rabbit Universal Secondary Antibody) | mIHC | Absin |
| abs50015 | 7-Color Multiplex Fluorescent IHC Kit (Mouse-Rabbit Universal Secondary Antibody) | mIHC | Absin |
| abs50028 | 4-Color Multiplex Fluorescent IHC Kit (Anti-Rabbit Secondary Antibody) | mIHC | Absin |
| abs50029 | 5-Color Multiplex Fluorescent IHC Kit (Anti-Rabbit Secondary Antibody) | mIHC | Absin |
| abs50030 | 6-Color Multiplex Fluorescent IHC Kit (Anti-Rabbit Secondary Antibody) | mIHC | Absin |
| abs50031 | 7-Color Multiplex Fluorescent IHC Kit (Anti-Rabbit Secondary Antibody) | mIHC | Absin |
ANT BIO PTE. LTD. – Empowering Scientific Breakthroughs
At ANTBIO, we are committed to advancing life science research through high-quality, reliable reagents and comprehensive solutions. Our specialized sub-brands (Absin, Starter, UA) cover a full spectrum of research needs, from general reagents and kits to antibodies and recombinant proteins. With a focus on innovation, quality, and customer-centricity, we strive to be your trusted partner in unlocking scientific mysteries and driving medical progress. Explore our product portfolio today and elevate your research to new heights.
Disclaimer
This article was partially created with the assistance of artificial intelligence. If any content involves copyright or intellectual property issues, please inform us, and we promise to verify and remove it immediately.




