Strategy for animal brain organoid culture
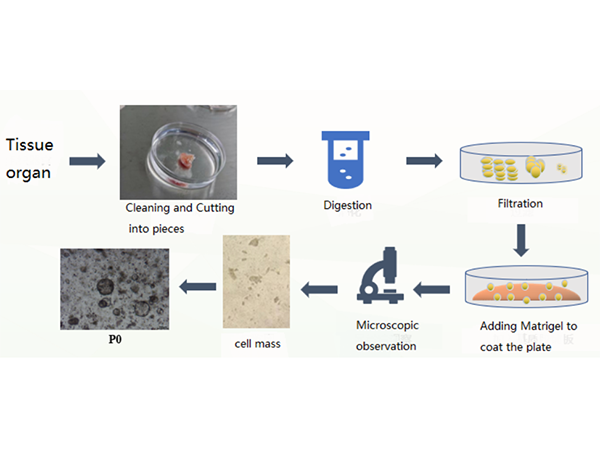
Process
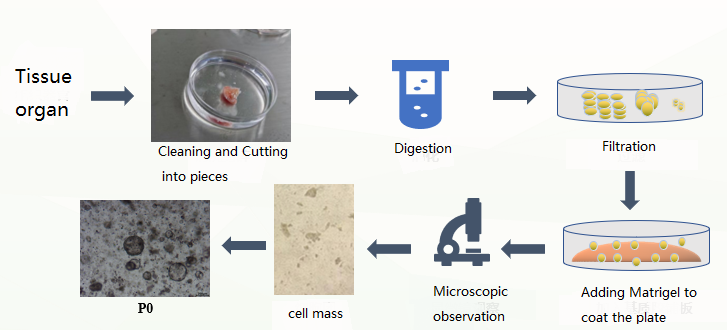
Organoid Primary Culture Process
Primary generation
01 Preparatory work
1. Instruments and equipment
CO2 incubator, double single-sided ultra-clean table, inverted microscope, desktop refrigerated centrifuge, water bath (abs72023) or water bath shaker, medical refrigerator,-80 ℃ refrigerator, pipette (set), ophthalmic scissors, ophthalmic forceps
2. Reagent consumables (taking intestinal cancer as an example)
Animal brain organoid culture medium kit (abs90050), Matrigel (low factor, no phenol red) (abs9495), 60mm cell culture dish (abs7005), 100μm sieve (abs7009), 15mL centrifuge tube (abs7102), several 1.5 mL EP tubes (abs7119), 24-well cell culture plate (abs7035), metal ice box, ophthalmic scissors, ophthalmic forceps

|
Component Name |
Specifications |
|
Animal Brain Organoid Medium A |
100mL |
|
Organoid primary culture buffer B |
250mL |
|
Primary tissue digestive juice C |
30mL |
|
Organoid passage digestive juice D |
30mL |
|
Tissue preservation solution E |
100mL |
|
Organoid Cryopreservation Solution F |
20mL |
|
Organoid Subculture Buffer G |
250mL |
02 Operation process
1. Sample preparation
(1) Place the tissue into a sampling bottle containing pre-cooled (2-8 °C) tissue preservation solution E (submerge the entire tissue) and retrieve it from the hospital/laboratory at 4 °C;
(2) Sterilize the sampling bottle, take out the tissue and put it into a petri dish sample to take photos, and register the information such as size, color, softness and hardness, tissue type, etc.
2. Cleaning-shredding
(1) Soak in a 60 mm cell culture dish (abs7005) with 2-3 mL of primary culture buffer B. After washing three times with primary culture buffer B (each time the dish was changed), the tissue pieces were cut into approximately 1-3 mm3 pieces and transferred to 15 mL centrifuge tubes.
3. Digestion-filtration
(1) Add 5 times primary tissue digestive juice C to a 15mL centrifuge tube (digestive fluid volume: tissue volume = 5: 1, if it is difficult to estimate the tissue volume, using 5mL digestive juice is usually enough) for digestion at 37 °C for 15-30min (Observe the digestion at any time during digestion).
(2) Take a small amount of liquid and observe it under a microscope. After more cell clusters (5-50 cell clusters) are observed under the microscope, add 3 times the volume of primary culture buffer B (buffer volume: digestion volume = 3: 1) to stop digestion, and you can see that the liquid becomes turbid by gently blowing with the gun tip.

(3) Filter using a 100 μm sieve (abs7009), and observe a small amount of the filtrate under a microscope. The filtrate was collected into a 15 mL centrifuge tube, and the supernatant was removed after enrichment centrifugation at 300 g at 4 °C for 5 min.
4. Add glue-spot board-add liquid (here is the finishing touch of the entire primary operation)
(1) Preparation
A Matrigel needs to be stored in a metal ice box at 4 ℃ refrigerator overnight to melt;
B The gun tip and centrifuge tube need to be pre-cooled at-20 ℃ for at least half an hour in advance;
C The melted Matrigel can be stored at 4 °c all the time, and it is recommended to use it up within 2 weeks.

Cryogenic metal ice box (abs7289)
(2) Vaccination requirements
24-well plate (abs7035), 25 uL matrigel cell mass mixture per well, 500-750 uL organoid culture medium.
(3) Seeding density
Density recommendation 1: Matrigel volume: cell mass precipitation volume = 25: 1 (if it is difficult to estimate the cell mass precipitation volume, adding 300uL of Matrigel is usually enough)
Density recommendation 2: 500 cell mass/25uL Matrigel (if you want to count seeding, you can refer to this density recommendation)
(4) Adding glue-spot plate
Add matrigel (abs9495) to the cell pellet, blow and mix evenly (do not blow fully, it is easy to produce bubbles), and then spot plate. The entire operation takes place on a metal ice box or ice. After the operation is skilled, add glue, mix evenly, and control the spotting plate within half a minute, which is conducive to maintaining the good fluency of the matrix glue.
(5) Adding liquid
Place the laid culture plate in a 37 °C incubator for 40-60 minutes to gel, and add 500-750 μL of organoid medium A for culture. About 10-14 days, most organoids have a diameter of 200um-500um, and can be passaged.

Decking density
Passaging (digestion is divided into two situations)
01 Passaging steps with a large number or large volume of organoids
1. Organoid collection and washing
(1) Collection: Aspirate the culture medium with a pipette, add about 1-2mL of 4 ℃ organoid subculture buffer G to each well, gently blow the matrigel, and collect it in a 15mL centrifuge tube (24-well plate, every 5 wells for a group).
(2) Washing: Add organoid subculture buffer G to 14mL (the more buffer, the more fully diluted the matrigel and the easier it is to remove), and let it stand at 4 ℃ for 40min or-20 ℃ for 5min (the purpose is to soften the matrigel. If the refrigerator has a strong heat preservation effect and shorten the freezing time. When finding a suitable freezing time, you can take out the centrifuge tube and shake it. If you can't see the matrigel, it means that it has cooled).
(3) Next, centrifuge the centrifuge tube for 300g, 4 °C, and centrifuge for 5min. After centrifugation, there are usually these two situations:
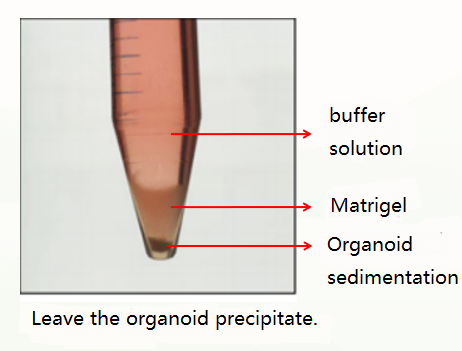
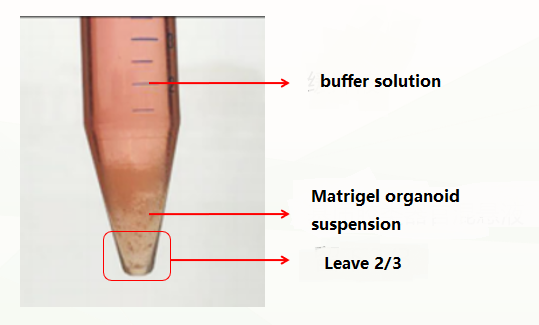
The first is the normal situation, which is divided into three layers (as shown in the figure below). At this time, the supernatant and matrigel layer are discarded, and the organoid precipitate is retained.
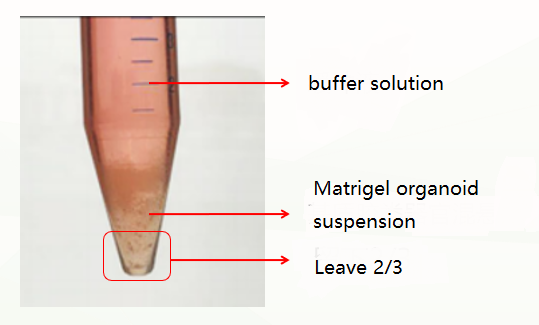
The second is an abnormal situation, which is divided into two layers (pictured below). This situation may be related to insufficient cooling. At this time, discard the buffer solution, leave the matrigel organoid suspension, repeat the previous washing step, and cold and centrifuge again. Usually, clear stratification (buffer layer, matrigel layer, organoid precipitate layer) can appear. At this time, discard the supernatant and matrigel layer, and keep the organoid precipitate. If there are still two layers (buffer layer and Matrigel organoid suspension layer), discard the buffer and the upper 1/3 of Matrigel organoid suspension at this time, and keep the lower 2/3.
Conditions to promote efficient separation of matrigel and organoids:
A The selection of centrifuge is very important. Compared with fixed angle centrifuge, horizontal angle centrifuge is more conducive to the separation of matrigel and organoid;
B The temperature of the centrifuge is preferably 4 ℃ (to avoid the curing of the matrigel), the centrifugation speed can be appropriately increased (the maximum should not exceed 500g), and the centrifugation time can be appropriately lengthened (the most common should not exceed 10min).
2. Organoid digestion
(1) Add 2-3mL of organoid passage digestive juice D and digest it in an ultra-clean table for 2-3min, and blow it 1-2 times during digestion. This step is mainly digested into cell clusters, and never digested into single cells. The survival rate of single-cell organoids is low. If you are not sure whether digestion is suitable, you can draw a few microliters and observe it under a microscope. If there are more cell masses, you can stop digestion.
(2) Add 5 times organoid subculture buffer G (buffer: digestion juice = 5: 1) to terminate digestion, centrifuge 300g at 4 ℃ for 5min and discard the supernatant (if there is matrigel residue, the residual amount < 50uL is normal, and does not affect the proliferation of subculture organoids)
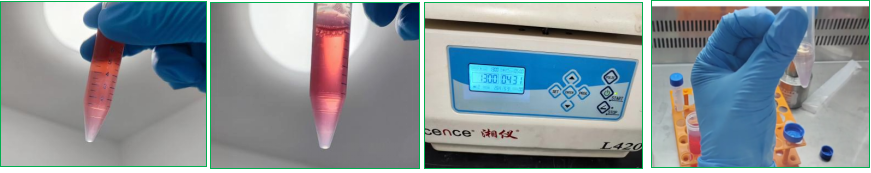
3. Add glue-spot plate-add liquid
(1) Preparation
A Matrigel needs to be stored in a metal ice box at 4 ℃ refrigerator overnight to melt;
B The gun tip and centrifuge tube need to be pre-cooled at-20 ℃ for at least half an hour in advance;
C The melted Matrigel can be stored at 4 °c all the time, and it is recommended to use it up within 2 weeks.
(2) Vaccination requirements
24-well plate (abs7035), 25 uL matrigel cell mass mixture per well, 500-750 uL organoid culture medium.
(3) Seeding density
Density Recommendation 1: Organoids are usually passaged at a ratio of 1: 2, for example, 5 wells are collected in a 24-well plate, 10 wells are passaged, and the amount of Matrigel required is 25 * 10 = 250uL
Density recommendation 2: 500 cell mass/25uL Matrigel (if you want to count seeding, you can refer to this density recommendation)
Note: Regardless of whether according to density recommendation 1 or density recommendation 2, if there is residual matrigel, the amount of new glue is at least 1.5 times the residual glue amount.
(4) Adding glue-spot plate
Add matrigel (abs9495) to the cell pellet, blow and mix evenly (do not blow fully, it is easy to produce bubbles), and then spot plate. The entire operation takes place on a metal ice box or ice. After the operation is proficient, add glue, mix evenly, and control the spotting plate within half a minute, which is conducive to maintaining the good fluency of the matrix glue.
(5) Adding liquid
The prepared culture plate was placed in a 37 °C incubator for 40-60min to gel, and 500-750μL of human intestinal cancer organoid medium A was added for culture. About 10-14 days, most organoids are 200-300um in diameter, and can be passaged.
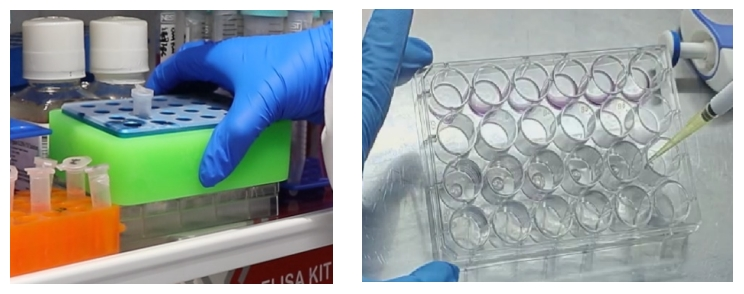
02 When the number of organoids is insufficient or the volume is small:
1. Organoid collection, pipetting and washing
(1) Aspirate the culture medium with a pipette, add about 1-2mL of 4 ℃ organoid subculture buffer G to each well, and gently blow away the matrigel;
(2) Perform pipetting and blow the organoids into cell clusters (pipetting can be stopped when there are more cell clusters observed under a sampling microscope);
(3) Washing: 24-well plate, each 5-well group, collected in a 15mL centrifuge tube, added organoid subculture buffer G to 14mL (the more buffer, the more fully diluted the matrigel, the easier it is to remove), and let it stand at 4 ℃ for 40min or-20 ℃ for 5min (the purpose is to soften the matrigel. If the refrigerator has strong heat preservation effect and shorten the freezing time.
(4) Next, centrifuge the centrifuge tube for 300g, 4 ℃, and centrifuge for 5min. After centrifugation, there are usually these two situations:
The first is the normal situation, which is divided into three layers (as shown in the figure below). At this time, the supernatant and matrigel layer are discarded, and the cell cluster precipitation can be retained.

The second is an abnormal situation, which is divided into two layers (pictured below). This situation may be related to insufficient cooling. At this time, discard the buffer solution, leave the matrigel organoid suspension, repeat the previous washing step, and cold and centrifuge again. Usually, clear stratification (buffer layer, matrigel layer, organoid precipitate layer) can appear. At this time, discard the supernatant and matrigel layer, and keep the organoid precipitate. If there are still two layers (buffer layer and matrigel cell mass suspension layer), discard the buffer and the upper 1/3 of the matrigel cell mass suspension at this time, and keep the lower 2/3.
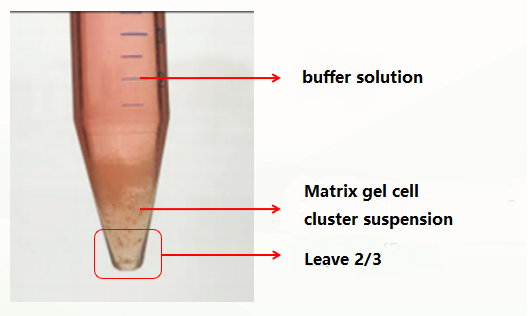
Conditions to promote efficient separation of matrigel and organoids:
A The selection of centrifuge is very important. Compared with fixed angle centrifuge, horizontal angle centrifuge is more conducive to the separation of matrigel and organoid;
B The temperature of the centrifuge is preferably 4 ℃ (to avoid the curing of the matrigel), the centrifugation speed can be appropriately increased (the maximum should not exceed 500g), and the centrifugation time can be appropriately lengthened (the most common should not exceed 10min).
2. Add glue-spot plate-add liquid
(1) Preparation
A Matrigel needs to be stored in a metal ice box at 4 ℃ refrigerator overnight to melt;
B The gun tip and centrifuge tube need to be pre-cooled at-20 ℃ for at least half an hour in advance;
C The melted Matrigel can be stored at 4 °c all the time, and it is recommended to use it up within 2 weeks.
(2) Vaccination requirements
24-well plate (abs7035), 25 uL matrigel cell mass mixture per well, 500-750 uL organoid culture medium.
(3) Seeding density
Density Recommendation 1: For a small number of organoids, in order to maintain the paracrine signal required for growth, 2-3 wells need to be enriched to 1 well. For example, 6 wells are collected in a 24-well plate, 1 well is enriched in 2 wells, and 3 wells are laid, and the amount of matrigel required is 25 * 3 = 75uL.
Density recommendation 2: 500 cell mass/25uL Matrigel (if you want to count seeding, you can refer to this density recommendation)
Note: Whether according to Density Recommendation 1 or Density Recommendation 2, if there is residual Matrigel, the amount of new glue is at least 1.5 times the amount of residual glue
(4) Adding glue-spot plate
Add matrigel (abs9495) to the cell pellet, blow and mix evenly (do not blow fully, it is easy to produce bubbles), and then spot plate. The entire operation takes place on a metal ice box or ice. After the operation is skilled, add glue, mix evenly, and control the spotting plate within half a minute, which is conducive to maintaining the good fluency of the matrix glue.
(5) Adding liquid
Place the laid culture plate in a 37 °C incubator for 40-60 minutes to gel, and add 500-750 μL of organoid medium A for culture. About 7-10 days, most organoids are 200-300um in diameter, and can be re-passaged.
Cryopreservation
Cryopreservation essentials:
Organoids do not require digestion (digested organoids have low resuscitative viability);
Cryopreservation during the exponential growth period of organoids (the activity of organoids is worse than that in the exponential period by the time of passage), that is, Day3-Day4 after passage, most organoids have a diameter of 100um-200um, and cryopreservation is selected at this time.
01 Organoid collection and washing
1. Collection: Aspirate the culture medium with a pipette, add about 1-2mL of 4 ℃ organoid subculture buffer G to each well, gently blow the matrigel, and collect it in a 15mL centrifuge tube (24-well plate, every 5 wells is a group).
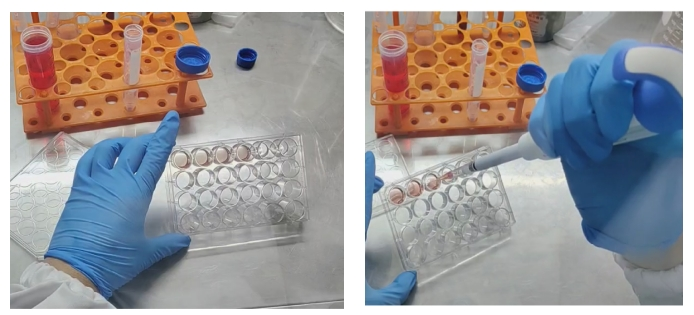
2. Washing: Add organoid subculture buffer G to 14mL (the more buffer, the more fully diluted the matrigel and the easier it is to remove), let it stand at 4 °C for 40min or-20 °C for 5min (the purpose is to soften the matrigel. If the refrigerator has a strong heat preservation effect and shorten the freezing time. When finding a suitable freezing time, you can take out the centrifuge tube and shake it. If you can't see the matrigel, it means that it has cooled).
3. Next, centrifuge the centrifuge tube for 300g at 4 °C for 5 minutes. After centrifugation, there are usually these two situations:
The first is the normal situation, which is divided into three layers (as shown in the figure below). At this time, the supernatant and matrigel layer are discarded, and the organoid precipitate is retained.

The second is an abnormal situation, which is divided into two layers (pictured below). This situation may be related to insufficient cooling. At this time, discard the buffer solution, leave the matrigel organoid suspension, repeat the previous washing step, and cold and centrifuge again. Usually, clear stratification (buffer layer, matrigel layer, organoid precipitate layer) can appear. At this time, discard the supernatant and matrigel layer, and keep the organoid precipitate. If there are still two layers (buffer layer and Matrigel organoid suspension layer), discard the buffer and the upper 1/3 of Matrigel organoid suspension at this time, and keep the lower 2/3.
Conditions to promote efficient separation of matrigel and organoids:
A The selection of centrifuge is very important. Compared with fixed angle centrifuge, horizontal angle centrifuge is more conducive to the separation of matrigel and organoid;
B The temperature of the centrifuge is preferably 4 ℃ (to avoid the curing of the matrigel), the centrifugation speed can be appropriately increased (the maximum should not exceed 500g), and the centrifugation time can be appropriately lengthened (the most common should not exceed 10min).
02 Cryopreservation of organoids
1. Cryopreservation density, taking 24-well plate as an example
Density recommendation 1: 2 wells/mL cryopreservation solution
Density recommendation 2: 500 organoids/mL cryopreservation solution (if you want to count cryopreservation, you can refer to this density recommendation)
2. Add an appropriate amount of organoid cryopreservation solution F, gently blow and resuspend, and it is recommended to cryopreserve immediately. (If left for too long, DMSO will damage organoids)
3. Gradient cryopreservation: Put the cryopreservation tube into the gradient cryopreservation box and store it to-80 °C overnight. Take it out the next day and put it into the liquid nitrogen tank.
Manual freezing: Place it in the refrigerator at 4 °C for 30 minutes, transfer it to-20 °C for 1 hour, then move it to-80 °C overnight, take it out the next day and put it in a liquid nitrogen tank.
Resuscitation
01 Preparation before the experiment
1. Preheat the water bath to 37 °C;
2. The cell laboratory is routinely disinfected, sprayed with preventive spray and irradiated with ultra-clean workbench surface for 40 minutes with ultraviolet rays;
3. Place the sterilized centrifuge tubes, straws, culture plates, etc. in order in the ultra-clean workbench.
02 Take out the cryopreservation tube
1. Find the number of the required organoid according to the label according to the cryopreservation record of the organoid;
2. Take out the cryopreservation box from the liquid nitrogen tank, take out the required cryopreservation tube, and check the number outside the cryopreservation tube.
03 Thaw quickly
1. Quickly put the cryopreservation tube into the preheated water bath to thaw, and shake it constantly to make the liquid in the tube melt quickly;
2. After about 1-2 minutes, the liquid in the cryopreservation tube is completely dissolved. Take it out and wipe the outer wall of the cryopreservation tube with an alcohol cotton ball, and then take it into the ultra-clean table.
04 Transfer the organoid cryopreservation solution into a 15mL centrifuge tube, add 10 times the volume of organoid subculture buffer G (buffer volume: cryopreservation volume = 10: 1) to resuspend, gently pig and mix well, 300g Centrifuge at 4 ℃ for 5 minutes, and discard the supernatant.
05 Adding glue-spot plate-adding liquid
1. Preparation work
(1) Matrigel should be stored in a metal ice box in a refrigerator at 4 ℃ overnight to melt;
(2) The gun tip and centrifuge tube need to be pre-cooled at-20 °C for at least half an hour in advance;
(3) The melted matrigel can be stored at 4 °C all the time, and it is recommended to use it up within 2 weeks.
2. Resuscitation requirements
24 well plate (abs7035), 25 uL of Matrigel organoid mixture per well, 500-750 uL of organoid culture medium.
3. Resuscitation density
The resuscitation density is recommended to be 1: 1: 1 inoculation (it turns out that a few holes will be resuscitated after freezing a few holes);
Resuscitation density recommendation 2: 250 organoids/25uL Matrigel (if you want to count the inoculation, you can refer to this density recommendation).
4. Add glue-spot board
Add matrigel (abs9495) to the organoid precipitate, blow and mix well (do not blow full, it is easy to produce bubbles), and then spot plate. The entire operation takes place on a metal ice box or ice. After the operation is skilled, add glue, mix evenly, and control the spotting plate within half a minute, which is conducive to maintaining the good fluency of the matrix glue.
5. Add liquid
Place the laid culture plate in a 37 °C incubator for 40-60 minutes to gel, and add 500-750 μL of organoid medium A for culture. About 10-14 days, most organoids have a diameter of 200um-500um, and can be passaged.
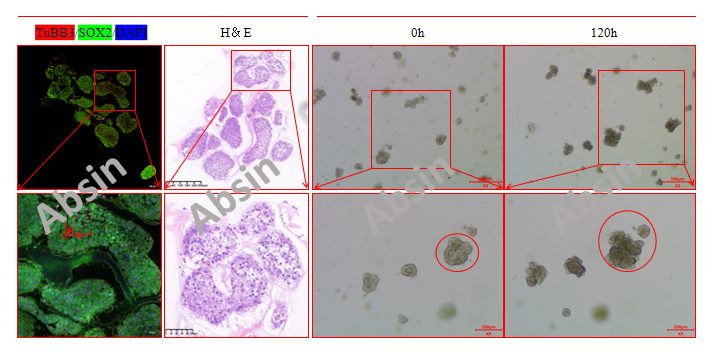
6. Brain organoid identification map
Recommended by Xiaoai in this issue
|
Item number |
Product name |
Specifications |
|
Organotial human gastric cancer organoid culture medium kit |
1kit |
|
|
Matrigel (low factor, no phenol red) |
1.5 ml * 8 |
|
|
abs7289 |
2mL cryogenic metal ice box (24 holes, flat bottom) |
1 |
|
abs7005 |
60mm cell culture dish |
1 box |
|
abs7233 |
100 μm filter screen |
1 box |
|
abs7164 |
Cell cryopreservation tube |
1 box |
|
abs7035 |
Cell culture plate (standard clear 24-well plate) |
1 box |
|
abs7119 |
1.5 mL EP Manage a few |
1 box |
|
abs7102 |
15mL centrifuge tube |
1 box |
|
abs7100 |
50mL centrifuge tube (pointed bottom) |
1 box |
|
abs7053 |
10mL disposable pipette |
1 box |
|
abs7054 |
25mL disposable pipette |
1 box |
Absin provides antibodies, proteins, ELISA kits, cell culture, detection kits, and other research reagents. If you have any product needs, please contact us.




