Storage Stability Test: A549 cells at a density of 1×10⁵ cells/mL were seeded into white 96-well plates with transparent bottoms at 100 µl per well; after 24 hours, the plates were equilibrated to room temperature, and UA-Glo Cell Viability Assay Reagent stored at room temperature for different durations was added at 50 µl per well and mixed well; fluorescence values were read after 10 minutes. The same batch of product stored at -20℃ was used as a control (0 hr), and the percentage of relative fluorescence values was calculated.
Product Details
Product Details
Product Specification
| Synonyms | Cell Viability Assay Kit |
| Stability & Storage |
Store at room temperature (22°C) for 1 week, at 4°C for 2 months, and at -20°C or below for 1 year. |
Background
The UA-Glo Homogeneous Ready-to-Use Cell Viability Assay Kit is used to quantitatively detect the content of ATP in living cells, thereby indirectly determining the number of living cells. This reagent features high signal-to-noise ratio, good repeatability, and excellent stability. The ready-to-use formula eliminates the experimental steps of first lysing and then detecting, thereby further reducing errors caused by frequent sample addition. Moreover, its stable glow signal makes this product particularly suitable for high-throughput tumor cell proliferation detection and compound screening.
Components
The product is filled into 10 ml or 100 ml brown bottles after mixing luciferase, luciferin, and buffer solution.
|
Product specifications |
Can detect the number of wells in a 96-well plate |
Can detect the number of wells in a 384-well plate |
|
1 x 10 ml |
200 |
1,000 |
|
1 x 100 ml |
2,000 |
10,000 |
|
10 x 100 ml |
20,000 |
100,000 |
Protocol
1.Cell Preparation
1) Seed the cells to be tested at an appropriate density in a 96-well or 384-well cell culture plate. It is recommended to use a white plate.
2) Prepare the test compounds. Add the test compounds at appropriate concentrations to the wells of the cell plate. The concentration of organic solvents in the culture medium should be kept below 1-2%. Set up a control group without added compounds. Continue culturing for an appropriate time according to the project requirements.
2. Cell Viability Assay
1) Take out the UA-Glo Cell Viability Assay Reagent and equilibrate it at room temperature for 20 minutes. Gently shake to mix well.
2) Take out the cell culture plate to be tested and equilibrate it at room temperature for 20 minutes.
3) Add 50 µl of the assay reagent to 100 µl of cells in a 96-well plate, or 10 µl of the reagent to 20 µl of cells in a 384-well plate, gently oscillate for 2 minutes, and place in the dark for further lysis for 10 minutes.
4) Read the luminescent signal on a luminescence microplate reader.
Guidelines
1. Temperature: Unless otherwise specified, luciferase detection is performed at room temperature (22-25°C). Temperature has a significant impact on enzyme reactions.
2. Reagent dosage: It is not recommended to arbitrarily change the dosage of reaction reagents without strict verification.
3. Plate reading time: After adding the reagents, shake for 2 minutes, place in the dark for 10 minutes, and then read the plate. It is recommended to complete the plate reading within 2 hours after adding the reagents.
4. Inter-plate internal reference: If inter-plate data comparison is required, it is recommended to set up two wells of positive controls without added compounds in all reaction plates as inter-plate internal references. The readings of each plate should first be corrected (normalization) using the inter-plate internal reference readings, and the corrected data should then be subjected to downstream processing and analysis.
5. Reaction plate: The level of fluorescence reading may significantly interfere with the readings of adjacent wells, mainly due to the spillover of fluorescence signals. When necessary, opaque-bottom white plates or black plates can be used for experiments or verification.
6. Application scenarios: This product is a detection reagent for live cells. If used in in vitro reaction systems constructed with recombinant proteins, additional ATP needs to be provided.
7. Reagent mixing: It is not recommended to mix reagents from different batches, reagents that have been opened but not used up, and reagents with significantly different storage conditions.
8. Aliquot storage: Aliquot and store the reaction reagents according to the instructions to ensure the stability of the reagents.
Picture
Picture
Bioactivity
Sensitivity Test: HEK293 cells subjected to two-fold serial dilution were seeded into white 96-well plates with transparent bottoms at 100 µl per well; after 24 hours, the plates were equilibrated to room temperature, and UA-Glo Cell Viability Assay Reagent (equilibrated to room temperature) was added at 50 µl per well and mixed well; fluorescence values were read after 10 minutes. There was a good linear relationship between fluorescence signal and cell number (r²>0.999), and as few as 100 cells per well could be detected.
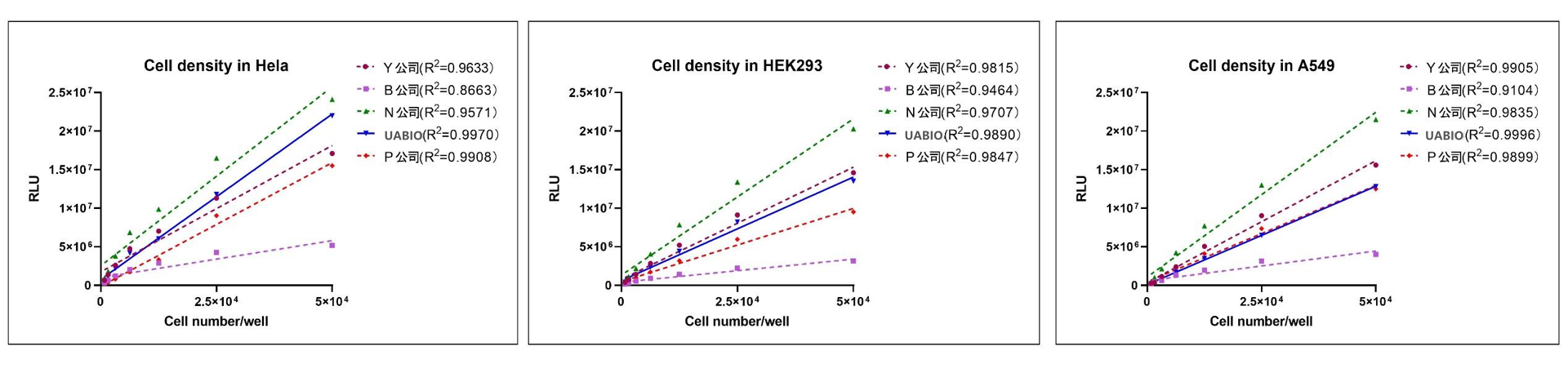
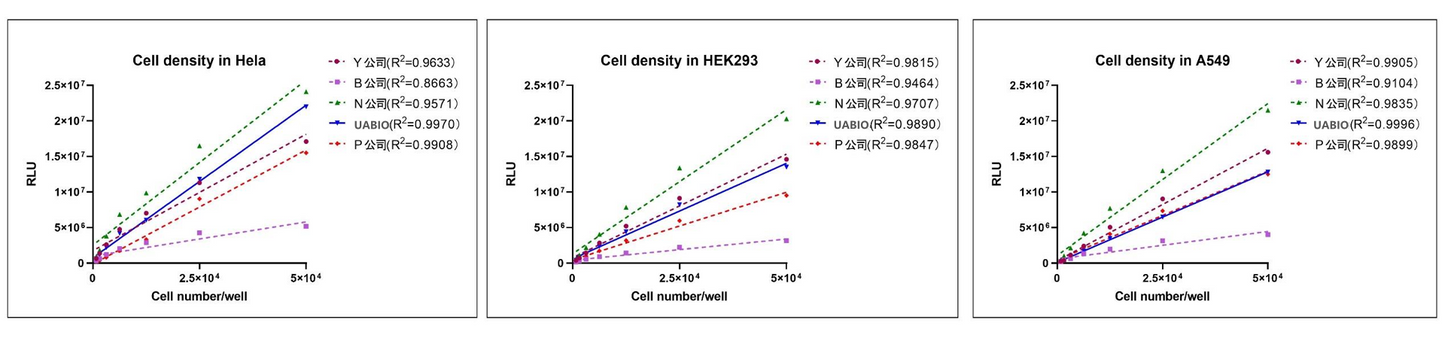
Linear Comparison with Competitor Products: Hela cells, HEK293 cells, and A549 cells were counted using a hemocytometer, and the cell concentration was adjusted to 5×10⁵ cells/mL. The serially two-fold diluted cells were seeded into white 96-well plates with transparent bottoms at 100 µl per well; UA-Glo Cell Viability Assay Reagent (equilibrated to room temperature), domestic competing reagents of the same type, and imported competing reagents of the same type (Company P) were added at 50 µl per well and mixed well; fluorescence values were read after 10 minutes. The results showed that UA-Glo Cell Viability Assay Reagent (Neubo) exhibited the optimal linearity among all three cell types.
Determination of Staurosporine Activity in Different Tumor Cells: A549 cells, HeLa cells, MCF-7 cells, and HCT116 cells at a density of 1–5×10⁴ cells/mL were seeded into white 384-well plates with transparent bottoms at 20 µl per well; after 24 hours, Staurosporine at different concentrations was added, and the cells were further cultured at 37℃ for 72 hours. The cells were then equilibrated to room temperature, and UA-Glo Cell Viability Assay Reagent (equilibrated to room temperature) was added at 10 µl per well and mixed well; fluorescence values were read after 10 minutes. The IC50 values of Staurosporine in A549 cells, HeLa cells, MCF-7 cells, and HCT116 cells were 2.9 nM, 5.4 nM, 16.5 nM, and 33 nM, respectively.
*: In the Z'-factor assay, each cell type at a certain concentration was seeded into a white 384-well plate with transparent bottoms at 20 µl per well. In Experiments 1, 2, and 3, 24 hours after cell plating, the left half of the plate was treated with 1 µM Staurosporine (final concentration of DMSO was 0.5%), and the right half was treated with 0.5% DMSO; in Experiments 4 and 5, 24 hours after cell plating, the right half of the plate was treated with 1 µM Staurosporine (final concentration of DMSO was 0.5%), and the left half was treated with 0.5% DMSO; 72 hours after the addition of Staurosporine and DMSO, the cells were equilibrated to room temperature, and UA-Glo Cell Viability Assay Reagent (equilibrated to room temperature) was added at 10 µl per well and mixed well; fluorescence values were read after 10 minutes.