UA-Glo Kinase ADP Max Assay: Standard curve of (ATP+ADP) mixture:
The (ATP+ADP) standard curve mixture was prepared according to the proportions in the above table: the maximum concentration of ADP and ATP in the 5mM standard curve mixture is 5mM.
Add 12.5μL of 5mM (ATP+ADP) standard curve mixture to each well of a 96-well white plate, then add 12.5μL of ATP MAX Removal Reagent. After incubating at room temperature for 40 minutes, add 25μL of ADP MAX Detection Reagent. Read the plate within 30 minutes to 180 minutes or longer; the standard curve is shown in the above figure.
Product Details
Product Details
Product Specification
| Synonyms | UA-Glo® Kinase ADP Max Assay Kit |
| Stability & Storage |
Transport on ice. Store at ≤ -20°C and protected from light, with a shelf life of 12 months. |
Background
The UA-Glo Kinase ADP-Max Assay Kit enables quantitative determination of the activity of any ADP-generating enzymes in enzymatic assays, such as kinases or ATPases. The kit determines enzyme activity by quantifying the amount of ADP, a product of the enzymatic reaction: the amount of ADP is positively correlated with enzyme activity. When using the UA-Glo Kinase ADP-Max Assay Kit, the maximum ATP concentration in the enzyme reaction can be up to 5 mM. The kit can detect most kinases, and kinase substrates include polypeptides, proteins, lipids, or sugars. This kit is suitable for high-throughput homogeneous screening of kinase/ATPase inhibitors. It can distinguish between ATP-competitive and non-competitive inhibitors by adjusting the ATP concentration. Moreover, using a high concentration of ATP in the experiment can make the reaction conditions of some kinases or ATPases more similar to their physiological conditions. The UA-Glo Kinase ADP-Max Assay Kit features broad versatility, high detection sensitivity, a wide detection dynamic range, and low susceptibility to compound interference.
Components
The Kinase ADP-Max Assay Kit consists of two components: ATP Max Depletion Reagent and ADP Max Detection Reagent. It also includes one vial of ATP Reagent and one vial of ADP Reagent respectively. The ATP Max Depletion Reagent is used to terminate the enzyme reaction and deplete ATP simultaneously. The ADP Max Detection Reagent is used to convert ADP into ATP, and generate a luminescent signal through the reaction between luciferase and ATP.
|
specs |
component |
Number of reactions in 96-well plates |
384-well plate reaction number |
|
1,000 assays |
ATP Max Depletion 5mL ADP Max Assay 10ml ATP (100mM, 0.25mL) ADP (10mM, 1mL) |
200 |
1,000 |
|
10,000 assays |
ATP Max Depletion 50mL ADP Max Assay100ml ATP (100mM, 2.5mL) ADP (10mM, 5mL) |
2,000 |
10,000 |
Protocol
1. Enzyme reaction experiment setup: Taking kinase reaction as an example
1) Perform the kinase reaction in a 96-well or 384-well white opaque assay plate. It is recommended that the reaction volume be 25μL for 96-well plates and 5μL for 384-well plates. Test compounds with concentration gradients can be added to the kinase reaction.
2) The concentrations of kinase and substrate need to be optimized according to different kinase reactions. Under the condition of the appropriate signal-to-noise ratio required for the experiment, the kinase concentration within the linear signal response range can be used for the experiment. Due to the high sensitivity of the UA-Glo Kinase ADP-Max Assay Kit, the amount of kinase used can be greatly reduced.
3) The concentration of ATP in the kinase reaction can be as high as 5mM, but not lower than 0.5mM. For kinase reactions with an ATP concentration lower than 0.5mM, it is recommended to use the UA-Glo Kinase ADP Assay Kit (Catalog No. UA070101) to obtain optimal experimental results. Some commercially available ATP contains residual ADP. Due to the high sensitivity of the Kinase ADP-Max Assay Kit, residual ADP in ATP will lead to high experimental background. Therefore, high-purity ATP must be used for the kinase reaction. The ATP provided with the kit can be used, or other high-purity ATP, such as ATP sold by Sigma-Aldrich (Cat# A2383, purity ≥99%) or ATP with higher purity.
4) The kinase reaction can be carried out in a universal reaction buffer (40mM Tris-HCl (pH 7.5), 0.1 mg/ml BSA, 20mM MgCl2) or using reaction buffers and cofactors reported in the literature.
5) The temperature and time of the kinase reaction need to be set according to different kinases. When conducting high-throughput screening experiments for compounds, it is recommended to optimize the kinase reaction to be carried out at room temperature (22℃-25℃) to facilitate maintaining the temperature uniformity of the assay plate during the Kinase ADP-Max detection process.
6) No additional reagents are needed to terminate the kinase reaction after it ends. If it is necessary to add a kinase reaction termination reagent due to special experimental requirements, avoid using magnesium ion chelators, such as EDTA. The Kinase ADP-Max detection reaction requires magnesium ions, and the final concentration of magnesium ions must be at least 5mM.
2. Removal of ATP after kinase reaction
1) Take out the ATP Max removal reagent and equilibrate it to room temperature (22℃-25℃). Gently shake to mix well [Note 1, 2, 3].
2) If the kinase reaction is carried out at a temperature other than room temperature, such as 30℃, equilibrate the assay plate to room temperature [Note 4].
3) Add 25 µL of ATP Max removal reagent to the above 25 µL 96-well assay plate (total volume 50 µL), or 5 µL of ATP Max removal reagent to the 5 µL 384-well assay plate (total volume 10 µL). Vortex to mix well [Note 5].
4) Incubate at room temperature for 40 minutes.
3. Kinase Activity Assay (ADP Detection)
1) Take out the ADP Max detection reagent and equilibrate to room temperature. Gently shake to mix [Note 4].
2) Add 50 µL of ADP Max detection reagent to the above 50 µL 96-well assay plate, or 10 µL of ADP Max detection reagent to the above 10 µL 384-well assay plate, and mix by shaking. Incubate at room temperature in the dark for 30 minutes.
3) The luminescent signal can be read 30-180 minutes or longer after adding the ADP Max detection reagent [Note 6].
Guidelines
1. After the first use of the reagent, it should be aliquoted and stored at -20°C or below in the dark to ensure the stability of the reagent. The ATP Max removal reagent and ADP Max detection reagent show no loss in signal intensity or functionality after 3 freeze-thaw cycles, and also no loss when stored at 4°C for 3 days.
2. The ADP Max detection reagent that has been stored at -20°C for a long time may have a small amount of precipitate when thawed to room temperature. The supernatant can be used directly or the precipitate can be removed by centrifugation before use.
3. Mixing of different batches is not recommended.
4. The luciferase reaction in the ADP Max detection reagent is sensitive to temperature changes. The reagent and test samples/assay plates need to be equilibrated to room temperature (22°C-25°C), and the temperature should be kept constant (±1°C) during the test.
5. It is not recommended to change the amount of reaction reagents unless verified. The volume ratio of the kinase reaction, ATP Max removal reagent, and ADP Max detection reagent should be 1:1:2.
6. The luminescent signal is very stable, with a decrease in signal intensity of <20% within 3 hours.
7. This product is for research use only.
Picture
Picture
Bioactivity
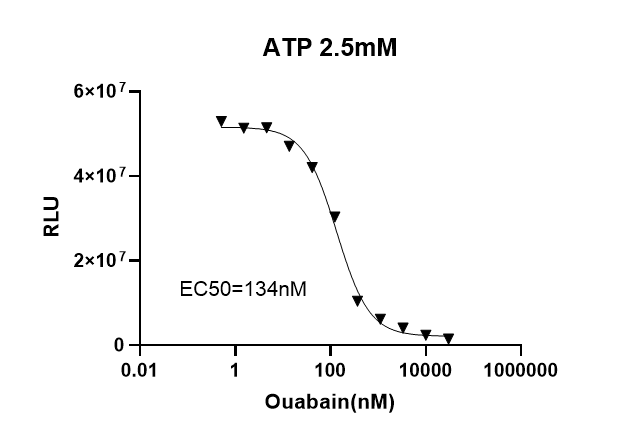
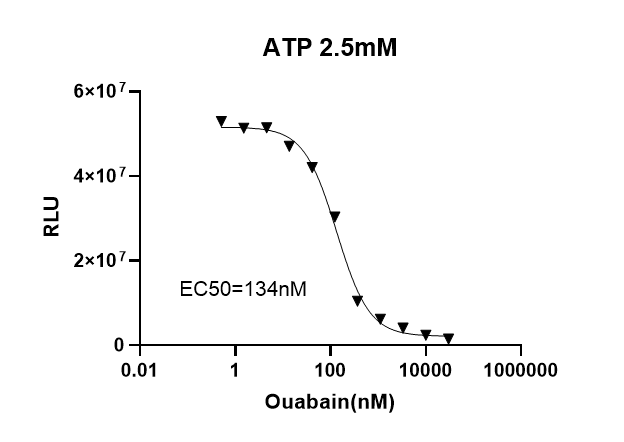
Determination of EC50 of Ouabain on Na⁺/K⁺ ATPase using UA-Glo Kinase ADP Max Detection Reagent:
Add 12.5μL of enzyme reaction solution (containing 2.5mM ATP, Na⁺/K⁺ ATPase, and Ouabain at different concentrations) to each well of a 96-well white plate, and incubate at 37°C for 1 hour. After equilibrating the assay plate to room temperature, add 12.5μL of ATP MAX Removal Reagent. After incubating at room temperature for 40 minutes, add 25μL of ADP MAX Detection Reagent. Read the plate within 30 minutes to 180 minutes or longer. The EC50 and dose-response curve are shown below; the measured EC50 is 134nM, which is consistent with the values reported in the literature.